计算物理 ›› 2021, Vol. 38 ›› Issue (3): 371-378.DOI: 10.19596/j.cnki.1001-246x.8253
• 研究论文 • 上一篇
收稿日期:2020-07-20
出版日期:2021-05-25
发布日期:2021-09-30
作者简介:Pan Jing (1979-), female, associate professor, research in photocatalytic properties of semiconductors, E-mail: jp@yzu.edu.cn
基金资助:
Jing PAN1( ), Guohua SHEN2
), Guohua SHEN2
Received:2020-07-20
Online:2021-05-25
Published:2021-09-30
摘要:
采用等价阴-阳离子共掺的方法调节ZnO的能带结构,提高其光催化分解水制氢的效率。计算结果表明:等价阴-阳离子共掺不仅减小了ZnO的带隙,使其在可见光区域的光吸收增强,而且能有效抑制电子-空穴的复合,提高载流子迁移率。(Cd+Te)共掺杂的ZnO是较理想的水分解的光催化剂,因为它具有合适的带隙、较少的电子-空穴复合中心、增强的可见光区域的光吸收和与水氧化还原势相匹配的带边位置。等价阴-阳离子共掺的方法也可运用到其它宽带隙的半导体中以提高光催化活性。
中图分类号:
潘靖, 沈国华. 等价阴-阳离子共掺杂调节ZnO的能带结构及其光催化活性[J]. 计算物理, 2021, 38(3): 371-378.
Jing PAN, Guohua SHEN. Enhanced Photocatalytic Activity of ZnO for Water-splitting with Isovalent Anion-Cation Codoping: First-principles Calculations[J]. Chinese Journal of Computational Physics, 2021, 38(3): 371-378.
| System | bond length | lattice | dipole | PBE-Eg | HSE-Eg | BGC | VBM | CBM | ||||
| Zn-O | Cd-O | Zn-S/Te/Se | a | c | ||||||||
| ZnO | 0.200 | 0.326 | 0.524 | 2.822 | 0.8 | 3.37 | -5.52 | -7.20 | -3.83 | |||
| Cd | 0.199 | 0.217 | 0.328 | 0.528 | 3.356 | 1.31 | 3.09 | |||||
| S | 0.200 | 0.223 | 0.329 | 0.531 | 1.502 | 1.78 | 3.34 | |||||
| Se | 0.200 | 0.232 | 0.330 | 0.534 | 1.175 | 1.68 | 3.18 | |||||
| Te | 0.201 | 0.245 | 0.332 | 0.537 | 0.514 | 1.49 | 2.81 | |||||
| (Cd+S) | 0.200 | 0.217 | 0.222 | 0.330 | 0.534 | 1.916 | 1.76 | 2.79 | -5.34 | -6.74 | -3.95 | |
| (Cd+Se) | 0.200 | 0.217 | 0.232 | 0.330 | 0.536 | 2.612 | 1.40 | 2.62 | -5.10 | -6.50 | -3.88 | |
| (Cd+Te) | 0.201 | 0.219 | 0.246 | 0.332 | 0.538 | 2.621 | 1.95 | 2.18 | -4.84 | -5.93 | -3.75 | |
Table 1 Average bond lengths (in nm), lattice parameters (in nm), dipole moments (in Debye), band gap (in eV) calculated with PBE method (PBE-Eg in eV) and HSE method (HSE-Eg in eV), calculated BGC, CBM and VBM (in eV) of pure, isovalent monodoped and codoped ZnO
| System | bond length | lattice | dipole | PBE-Eg | HSE-Eg | BGC | VBM | CBM | ||||
| Zn-O | Cd-O | Zn-S/Te/Se | a | c | ||||||||
| ZnO | 0.200 | 0.326 | 0.524 | 2.822 | 0.8 | 3.37 | -5.52 | -7.20 | -3.83 | |||
| Cd | 0.199 | 0.217 | 0.328 | 0.528 | 3.356 | 1.31 | 3.09 | |||||
| S | 0.200 | 0.223 | 0.329 | 0.531 | 1.502 | 1.78 | 3.34 | |||||
| Se | 0.200 | 0.232 | 0.330 | 0.534 | 1.175 | 1.68 | 3.18 | |||||
| Te | 0.201 | 0.245 | 0.332 | 0.537 | 0.514 | 1.49 | 2.81 | |||||
| (Cd+S) | 0.200 | 0.217 | 0.222 | 0.330 | 0.534 | 1.916 | 1.76 | 2.79 | -5.34 | -6.74 | -3.95 | |
| (Cd+Se) | 0.200 | 0.217 | 0.232 | 0.330 | 0.536 | 2.612 | 1.40 | 2.62 | -5.10 | -6.50 | -3.88 | |
| (Cd+Te) | 0.201 | 0.219 | 0.246 | 0.332 | 0.538 | 2.621 | 1.95 | 2.18 | -4.84 | -5.93 | -3.75 | |
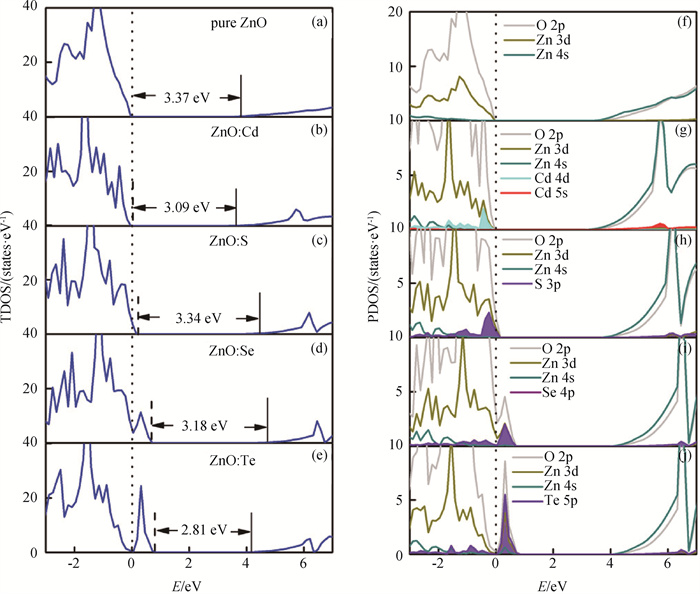
Fig.1 HSE-calculated (a)-(e) TDOS and (f)-(j) PDOS of pure, Cd-, S-, Se- and Te-doped ZnO (Fermi levels of systems are displayed with dashed lines. The dotted lines indicate the VBM of pure ZnO. The PDOS at conduction band are amplified for clarity.)
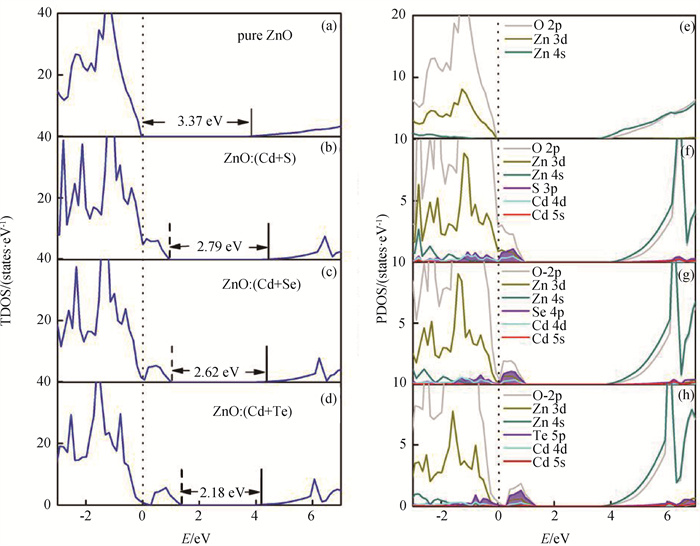
Fig.2 HSE-calculated (a)-(d) TDOS and (f)-(j) PDOS of pure, (Cd+S), (Cd+Se) and (Cd+Te)-codoped ZnO (Fermi levels of the systems are displayed with dashed lines. The dotted lines indicate the VBM of pure ZnO. The PDOS at conduction band are amplified for clarity.)
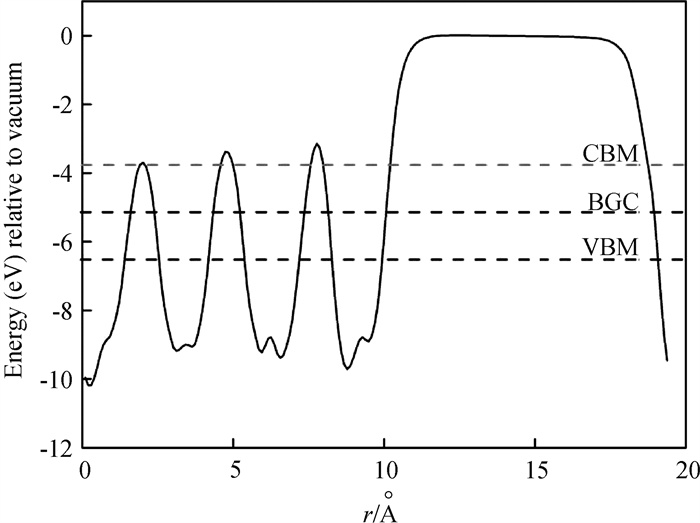
Fig.3 Electrostatic potential V(r) of the (10${\rm{\bar 1}}$0) surface of (Cd+Se) codoped ZnO (The vacuum level corresponds to zero electrostatic potential energy. The dash lines denote
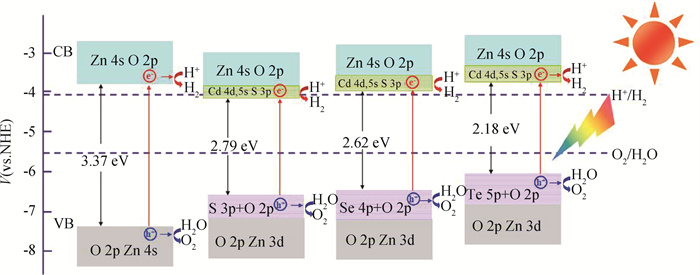
Fig.4 Band edges of pure and (Cd+S), (Cd+Se), (Cd+Te) codoped ZnO in comparison with redox potential of water at pH=7 (The introduction of dopants creates new energy levels that effectively narrow band gap.)
| 1 |
FUJISHIMA A, HONDA K. Electrochemical photolysis of water at a semiconductor electrode[J]. Nature, 1972, 238, 37- 38.
DOI |
| 2 |
GRAZEL M. Photoelectrochemical cells[J]. Nature, 2001, 414, 338- 344.
DOI |
| 3 | KUBACKA A, FERNANDEZ-GARCIA M, COLON G. Advanced nanoarchitectures for solar photocatalytic applications[J]. Chem Rev, 2012, 112, 1555- 1614. |
| 4 |
SU T, SHAO Q, QIN Z, et al. Role of interfaces in two-dimensional photocatalyst for water splitting[J]. ACS Catalysis, 2018, 8 (3): 2253- 2276.
DOI |
| 5 |
ASAHI R, MORIKAWA T, OHWAKI T, et al. Visible-light photocatalysis in nitrogen-doped titanium oxides[J]. Science, 2001, 293, 269- 271.
DOI |
| 6 |
YIN W J, TANG H, WEI S H, et al. Band structure engineering of semiconductors for enhanced photoelectrochemical water splitting: The case of TiO2[J]. Phys Rev B, 2010, 82, 045106.
DOI |
| 7 |
QIU Y, YAN K, DENG H, et al. Secondary branching and nitrogen doping of ZnO nanotetrapods: Building a highly active network for photoelectrochemical water-splitting[J]. Nano Lett, 2012, 12, 407- 413.
DOI |
| 8 |
THOMAS M A, CUI J B. Electrochemical route to p-type doping of ZnO nanowires[J]. J Phys Chem Lett, 2010, 1, 1090- 1094.
DOI |
| 9 |
YANG X, WOLCOTT A, WANG G, et al. Nitrogen-doped ZnO nanowire arrays for photoelectrochemical water splitting[J]. Nano Lett, 2009, 9, 2331- 2336.
DOI |
| 10 |
POORNAPRAKASH B, CHALAPATHI U, KUMAR M, et al. Enhanced photocatalytic degradation and hydrogen evolution of ZnS nanoparticles by (Co, Er) co-doping[J]. Matter Lett, 2020, 273, 127887.
DOI |
| 11 |
WEI S H. Overcoming the doping bottleneck in semiconductors[J]. Comput Mater Sc, 2004, 30, 337- 348.
DOI |
| 12 |
AHN K S, YAN Y, AL-JASSIM M. Band gap narrowing of ZnO: N films by varing rf sputtering power in O2/N2 mixtures[J]. J Vac Sci Technol B, 2007, 25, L23.
DOI |
| 13 |
JOUNG Y, LEE K, PARK M, et al. Electrical and optical properties of Al-doped ZnO transparent conductive oxide films prepared via radio frequency magnetron Co-sputtering system[J]. Journal of Nanoscience and Nanotechnology, 2020, 20 (11): 6788- 6791.
DOI |
| 14 |
JAKANI M, COMPET G, CLAVERIE J, et al. Photoelectrochemical properties of zinc oxide doped with 3d elements[J]. J Solid State Chem, 1985, 56, 269- 277.
DOI |
| 15 |
BAHADUR L, RAO T N. Photoelectrochemical studies of cobalt-doped ZnO sprayer thin film semiconductor electrodes in acetonitrile medium[J]. Sol Energy Mater Sol Cells, 1992, 27, 347- 360.
DOI |
| 16 |
ZHU W, QIU X, IANCU V, et al. Band gap narrowing of titanium oxide semiconductors by noncompensated anion-cation codoping for enhanced visible-light photoactivity[J]. Phys Rev Lett, 2009, 103, 226401.
DOI |
| 17 | GAI Y, LI J, LI S S, et al. Design of narrow-gap TiO2: A passivated codoping approach for enhanced photoelectrochemical activity[J]. Phys Rev Lett, 2009, 102, 0364021. |
| 18 |
PAN J, WANG S D, CHEN Q, et al. Band structure engineering of ZnO by anion-cation codoping for enhanced photoelectrochemical activity[J]. Chem Phys Chem, 2014, 15, 1611- 1618.
DOI |
| 19 |
MAYER M A, YU K M, SPEAKS D T, et al. Band gap engineering of oxide photoelectrodes: Characterization of ZnO1-xSex[J]. J Phys Chem C, 2012, 116, 15281- 15289.
DOI |
| 20 |
LAI H H C, KUZNETSOV V L, EGDELL R G, et al. Electronic structure of ternary CdxZn1-xO (0 ≤ x ≤ 0.075) alloys[J]. Appl Phys Lett, 2012, 100, 072106.
DOI |
| 21 | CHEN H X, DU S J, ZHUANG G C. Structure and magnetic properties of Ni-doped ZnO clusters[J]. Chinese J Comput Phys, 2018, 35 (1): 112- 118. |
| 22 | CHEN C T, CONG S, CHEN H F, et al. First-principles study of electronic structure and optical properties of Bi doped ZnO[J]. Chinese J Comput Phys, 2018, 35 (6): 720- 728. |
| 23 | LI L L, LI W X, DAI J F, et al. Effects of Zinc vacancies on electronic structure of Al-P co-doped ZnO: First-principles calculations[J]. Chinese J Comput Phys, 2017, 34 (6): 714- 721. |
| 24 | XIE J M, CHEN H X. Structure and magnetic properties of Co-doped (ZnO)12 clusters[J]. Chinese J Comput Phys, 2014, 31 (3): 372- 378. |
| 25 |
KRESSE G, FURTHMULLER J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set[J]. Phys Rev B, 1996, 54, 11169.
DOI |
| 26 |
VENKATESAN M, FITZGERALD C B, LUNNEY J G, et al. Anisotropic ferromagnetism in substituted zinc oxide[J]. Phys Rev Lett, 2004, 93, 177206.
DOI |
| 27 |
LI L, CUI X Y, OHTANI K, et al. Magnetism of co-doped ZnO epitaxially grown on a ZnO substrate[J]. Phys Rev B, 2012, 85, 174430.
DOI |
| 28 |
PERDEWJ P, BURKE K, ERNZERHOF M. Generalized gradient approximation made simple[J]. Phys Rev Lett, 1996, 77, 3865.
DOI |
| 29 |
OBA F, TOGO A, TANAKA I. Defect energetics in ZnO: A hybrid Hartree-Fock density functional study[J]. Phys Rev B, 2008, 77, 245202.
DOI |
| 30 |
HEYD J, SCUSERIA G E, ERNZERHOF M. Hybrid functionals based on a screened Coulomb potential[J]. J Chem Phys, 2006, 124, 219906.
DOI |
| 31 |
SATO J, KOBAYASHI H, INOUE Y. Photocatalytic activity for water decomposition of indates with octahedrally coordinated d 10 configuration II. Roles of geometric and electronic structures[J]. J Phys Chem B, 2003, 107, 7970- 7975.
DOI |
| 32 |
REYNOLDS D C, LOOK D C, JOGAI B, et al. Valence-band ordering in ZnO[J]. Phys Rev B, 1999, 60, 2340.
DOI |
| 33 |
HUTCHINSON D A W. Self-consistent effects of continuous wave output coupling of atoms from a Bose-Einstein condensate[J]. Phys Rev Lett, 1999, 82, 6.
DOI |
| 34 |
KANAN D K, CARTER E A. Band gap engineering of MnO via ZnO alloying: A potential new visible-light photocatalyst[J]. J Phys Chem C, 2012, 116, 9876- 9887.
DOI |
| 35 |
AKYOL A, YATMAZ H C, BAYRAMOGLU M. Photocatalytic decolorization of remazol red RR in aqueous ZnO suspensions[J]. Appl Cata B: Environ, 2004, 54, 19- 24.
DOI |
| [1] | 赵一程, 郭俊宏, 胡芳仁. 应变对单层砷烯结构拉曼散射的影响[J]. 计算物理, 2020, 37(3): 365-370. |
| [2] | 熊宗刚, 杜娟, 张现周. 二维GeSe纳米片五族和七族原子掺杂的受主和施主杂质态[J]. 计算物理, 2019, 36(6): 733-741. |
| [3] | 李永, 李海生, 李冠亚, 王赵武, 李国岭, 左正伟, 李立本. 合金效应加强水分子在PtRun团簇上的吸附作用[J]. 计算物理, 2017, 34(2): 230-236. |
| [4] | 张若兴, 侯士敏, 丑强. 基于第一性原理量子输运模拟的并行计算[J]. 计算物理, 2015, 32(6): 631-638. |
| [5] | 陈金繁, 罗超, 敖冰云, 彭丽霞, 石洁. V-Ta合金力学行为的计算与实验研究[J]. 计算物理, 2014, 31(5): 609-616. |
| [6] | 李祥然, 李丹, 王春雷, 牛原, 赵红敏, 梁春军. F原子吸附TiO2:Mn(001)稀磁半导体薄膜电子结构和磁性的第一性原理计算[J]. 计算物理, 2014, 31(1): 96-102. |
| [7] | 张芳英. 0~15GPa外压下ZnO结构相变的第-性原理研究[J]. 计算物理, 2012, 29(2): 303-307. |
| [8] | 李磊, 李丹, 刘世勇, 赵翼. Mn掺杂的ZnS(001)表面的电子态特性[J]. 计算物理, 2010, 27(2): 293-298. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
版权所有 © 《计算物理》编辑部
地址:北京市海淀区丰豪东路2号 邮编:100094 E-mail:jswl@iapcm.ac.cn
本系统由北京玛格泰克科技发展有限公司设计开发
